
The dipole moment is an important property to consider in various chemical reactions and physical interactions involving polar molecules. In conclusion, the dipole moment of water (H2O) is greater than that of ammonia (NH3) due to the higher electronegativity difference and shorter distance between charges in water. In ammonia, the nitrogen atom is less electronegative than oxygen, resulting in a larger distance between the charges and a smaller dipole moment compared to water. This results in a shorter distance between the positive and negative charges, further enhancing the dipole moment. In water, the oxygen atom is more electronegative, causing the electron density to be pulled closer to the oxygen atom. The dipole moment is also affected by the distance between the charges. PH3 has a lone pair and does not have a trigonal planar geometry-for this reason it is not symmetrical. PH3 must be polar since it is not symmetrical. Here is why: PH3 is called phosphine and it is quite toxic and flammable. The hydrides of group 5A are NH3, PH3, AsH3, and SbH3. It forms dipole-dipole because it is a polar molecule. Step 3: The boiling point of a compound increases with an increase in molar mass. Highest to lowest - SbH3, AsH3, PH3, and NH3. This greater electronegativity difference in water leads to a greater charge separation, resulting in a larger dipole moment compared to ammonia. Arrange the group 5A hydrides PH3, SbH3, NH3, and AsH3 from highest to lowest molar mass. In water (H2O), the electronegativity difference between oxygen and hydrogen atoms is higher than the electronegativity difference between nitrogen and hydrogen atoms in ammonia (NH3). The dipole moment of a molecule is influenced by two factors: the magnitude of the charge separation and the distance between the charges. 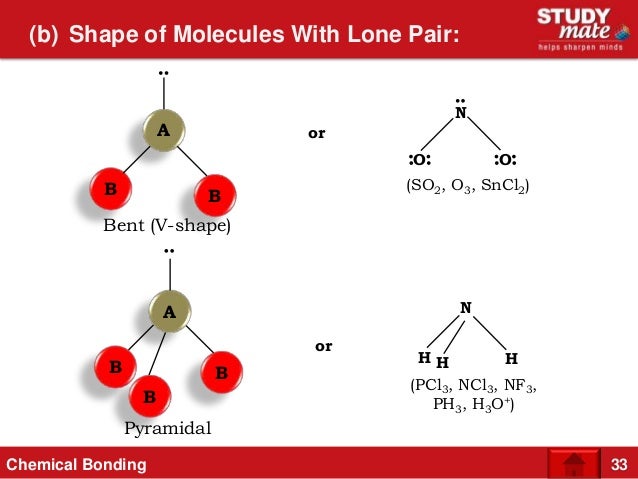
This separation of charges creates a dipole moment in the molecule. The nitrogen atom is more electronegative than the hydrogen atoms, leading to a partial negative charge on the nitrogen atom and partial positive charges on the hydrogen atoms. Similarly, ammonia is also a polar molecule due to its pyramidal molecular geometry and the electronegativity difference between nitrogen and hydrogen atoms. The oxygen atom is more electronegative than the hydrogen atoms, resulting in a partial negative charge on the oxygen atom and partial positive charges on the hydrogen atoms. 
Water is a polar molecule due to its bent molecular geometry and the electronegativity difference between oxygen and hydrogen atoms. In this case, we are comparing the dipole moments of water (H2O) and ammonia (NH3). A dipole moment occurs when there is a separation of positive and negative charges in a molecule. It is defined as the product of the charge (Q) and the distance (d) between the charges in a molecule. If the geometry is not totally symmetrical around its center, the molecule will be polar.The dipole moment is a measure of the polarity of a molecule. When one or more lone pairs are present on a central atom, the VSEPR model (correctly) predicts the geometry. The accepted Pauling values are 2.20 for H and 2.19 for P - which are very close to one another, but not the same.įinally, the geometry of a molecule is usually what dictates whether or not a molecule is polar. Secondly, the Pauling numbers that you are citing are rounded off to one decimal place. First of all, different EN scales exist besides the Pauling scale, and in those scales P and H do not have the same EN values as one another. The electronegativity (EN) concept is powerful, but the numbers that you use should not be taken too literally. Maybe you will not be able to view the article, but the abstract states that the measured dipole moment was 0.5740 ☐.0002 Debye. 
The dipole moment of phosphine is 0.58D which is less than 1.42D for NH3. PH3 is called phosphine and it is quite toxic and flammable. It forms dipole-dipole because it is a polar molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
